Projects
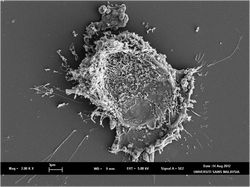
Anti-proliferation properties of H. brasiliensis latex sera
Latex from the natural rubber tree known as Hevea brasiliensis has always been considered as an economic commodity and never been thought as a medicinal alternative until recently. Our recent findings have indicated that latex B- and C- sera possessed anti-proliferation properties against specific human cancer-origin cell lines. The study has confirmed that latex C-serum fractions, DCS and DCP, were able to exert its anti-proliferation properties against cancer-origin cell lines and the cell death mechanism has been identified. DCS- and DCP-treated HepG2, MCF-7 and MDA-MB231 cells were positively-stained with propidium iodide and negatively-stained with Annexin V, indicating apoptosis might not be the mechanism of cell death. Cell cycle analysis using BrdU incorporation showed that the treated cells were arrested in G0/G1 phase compared to the non-treated cells. Furthermore, ultrastructural changes of DCS- and DCP-treated cells observed using SEM and TEM confirmed the cell death mechanism triggered as autoschizis. In addition to its potential medical use in cancer treatment, DCS fraction could be used to study the molecular signaling pathway(s) involved in autoschizis. Work is being conducted in animal model to explore the potential of the fractions in cancer treatment.
Latex from the natural rubber tree known as Hevea brasiliensis has always been considered as an economic commodity and never been thought as a medicinal alternative until recently. Our recent findings have indicated that latex B- and C- sera possessed anti-proliferation properties against specific human cancer-origin cell lines. The study has confirmed that latex C-serum fractions, DCS and DCP, were able to exert its anti-proliferation properties against cancer-origin cell lines and the cell death mechanism has been identified. DCS- and DCP-treated HepG2, MCF-7 and MDA-MB231 cells were positively-stained with propidium iodide and negatively-stained with Annexin V, indicating apoptosis might not be the mechanism of cell death. Cell cycle analysis using BrdU incorporation showed that the treated cells were arrested in G0/G1 phase compared to the non-treated cells. Furthermore, ultrastructural changes of DCS- and DCP-treated cells observed using SEM and TEM confirmed the cell death mechanism triggered as autoschizis. In addition to its potential medical use in cancer treatment, DCS fraction could be used to study the molecular signaling pathway(s) involved in autoschizis. Work is being conducted in animal model to explore the potential of the fractions in cancer treatment.
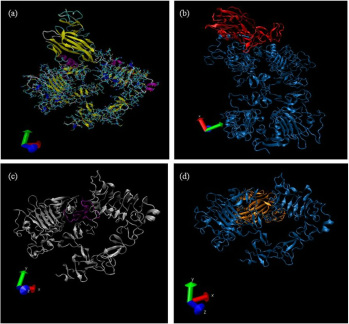
Study on the involvement of WIF1 in initiating oral carcinogenesis via EGFR signaling pathway
All protein-protein interaction is a well-known phenomenon in cancer cell signaling pathway. To study the potential docking between WIF-1 and EGFR molecules, initial protein-protein docking was performed using CAPRI-listed online servers, namely ZDOCK, GRAMM-X, HEX and PatchDock. The predicted docking conformation issued from various servers suggested the similar potential protein-protein conformational interaction. The lowest docking energy of WIF-1/EGFR binding calculated using HEX server was -616.40 kcal/mol. This was comparable to that of EGF/EGFR binding (-627.18 kcal/mol), indicating a possibility for WIF-1 to bind to EGFR through its EGF-like domain. In addition, we further confirmed the in silico findings using co-immunoprecipitation approach.
The capability of WIF-1 to act as a ligand to EGFR and trigger carcinogenesis or metastasis might explain the observations wherein the expression of WIF-1 was high in cancer cases (Suzuki et al. 2007a; Suzuki et al. 2007b). In these cases, WIF-1 might be playing dual roles by inhibiting WNT signaling pathway on one hand, but initiate EGFR signaling pathway on the other hand. This latter would engrave the situation by promoting cancer cell growth, while researchers are expecting WIF-1 being playing a cancer-cell-growth inhibiting role. The confirmation of the “bad guy” role of WIF-1 under the camouflage of it as inhibitor of WNT signaling pathway would pave new insights to the cancer formation and eventually assist in cancer treatment research. Current activities are focused on the activation of beta-catenin and the downstream activities following the activation of beta-catenin via phosphorylation, including identification of control element of downstream gene expression.
All protein-protein interaction is a well-known phenomenon in cancer cell signaling pathway. To study the potential docking between WIF-1 and EGFR molecules, initial protein-protein docking was performed using CAPRI-listed online servers, namely ZDOCK, GRAMM-X, HEX and PatchDock. The predicted docking conformation issued from various servers suggested the similar potential protein-protein conformational interaction. The lowest docking energy of WIF-1/EGFR binding calculated using HEX server was -616.40 kcal/mol. This was comparable to that of EGF/EGFR binding (-627.18 kcal/mol), indicating a possibility for WIF-1 to bind to EGFR through its EGF-like domain. In addition, we further confirmed the in silico findings using co-immunoprecipitation approach.
The capability of WIF-1 to act as a ligand to EGFR and trigger carcinogenesis or metastasis might explain the observations wherein the expression of WIF-1 was high in cancer cases (Suzuki et al. 2007a; Suzuki et al. 2007b). In these cases, WIF-1 might be playing dual roles by inhibiting WNT signaling pathway on one hand, but initiate EGFR signaling pathway on the other hand. This latter would engrave the situation by promoting cancer cell growth, while researchers are expecting WIF-1 being playing a cancer-cell-growth inhibiting role. The confirmation of the “bad guy” role of WIF-1 under the camouflage of it as inhibitor of WNT signaling pathway would pave new insights to the cancer formation and eventually assist in cancer treatment research. Current activities are focused on the activation of beta-catenin and the downstream activities following the activation of beta-catenin via phosphorylation, including identification of control element of downstream gene expression.

Anti-cancer properties of medicinal plants
We are interested in studying the anti-cancer properties of plants used in local traditional medicine. Most of the medicinal plants claimed to have anti-cancer properties are lack of proper scientific data for the known properties are principally based on empirical observations. It is therefore important to verify and elucidate these properties by using proper scientific means. We are currently conducting studies using extracts from Selaginella Doederleinii Hieron, Citrus hystrix, Nepenthes gracilis, and Crescentia cujete.
We are interested in studying the anti-cancer properties of plants used in local traditional medicine. Most of the medicinal plants claimed to have anti-cancer properties are lack of proper scientific data for the known properties are principally based on empirical observations. It is therefore important to verify and elucidate these properties by using proper scientific means. We are currently conducting studies using extracts from Selaginella Doederleinii Hieron, Citrus hystrix, Nepenthes gracilis, and Crescentia cujete.
